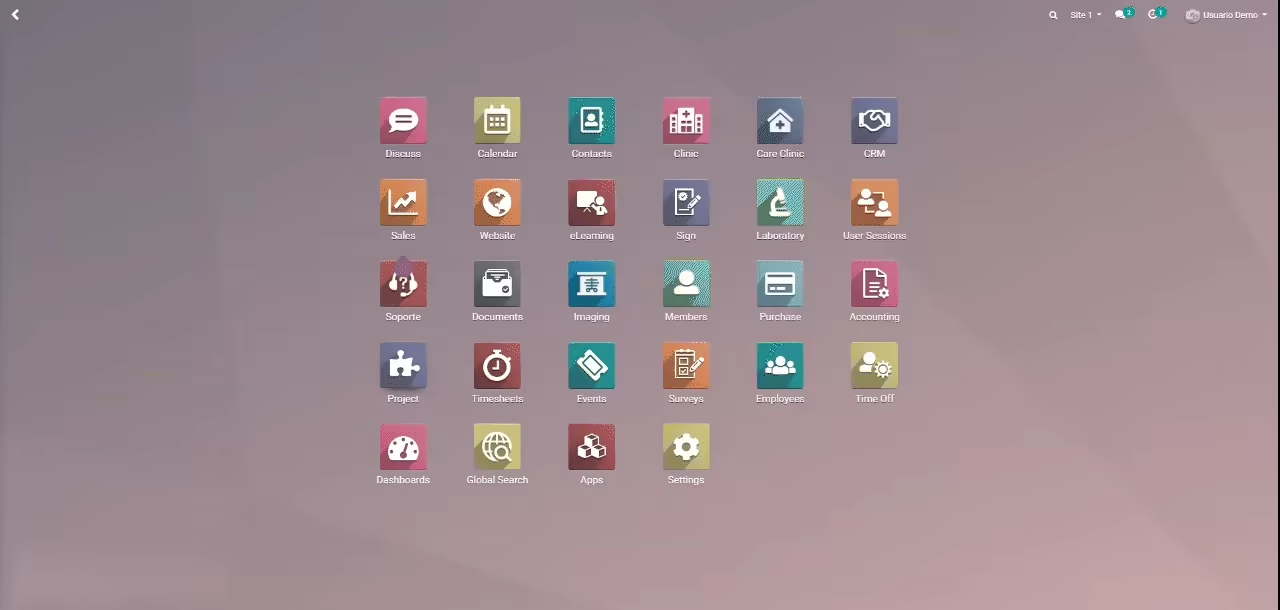
Trial360 is a web-based 21 CFR Part 11 compliant platform that speeds up processes and improves quality across the clinical trial. Inspired by a site’s network, this modular and user-friendly solution adapts to any site size as well as various studies for the investigational site at the same time.This tool has been used in sites to support multi-study, multi-site and multi-country organizations. Featuring several core modules: Research Projects, Recruitment, Scheduling, eSource: electronic medical record (EMR), Laboratory, pharmacy, Imaging. This integrated system allows in an organized and stress free manner investigational sites to comply with clinical practice guidelines.
Grow your sites even faster by installing the Enterprise Resource Planning (ERP) modules such as Finance and Billing, Procurement and Inventory, Human Resources, Document Management, eLearning, etc., on an on-demand basis and with a pay-as-you-go model.






100% Satisfactory FDA Inspections

Quick Project Set Up (By Project)

Recruitment (CRM - Website)

Management (Visits & eSource)

Data Analysis

This module is the forefront of your site; it allows you to manage multiple trials and sites, and establish protocols within hours, rather than days. This easy-to-setup system offers complete control and customization of all trial variables and centralizes all the data, such as visits, recruiting roles, and participants.
Main features:
With this module, your site can build a prospect subject database with detailed contact information, schedule follow-ups, and keep a record of the subject’s status until the last visit. Keep track of KPIs such as dropout motives, referrals, calls, and appointments in real-time.
Main features:
Turn complexity into ease of use with this module; setup, plan, program, and follow up all visits to your site. This user-friendly module optimizes resources improving appointment planning and patient flows according to the protocol, making sites more efficient with increased assistance rates and reduced dropout rates.
Main features:
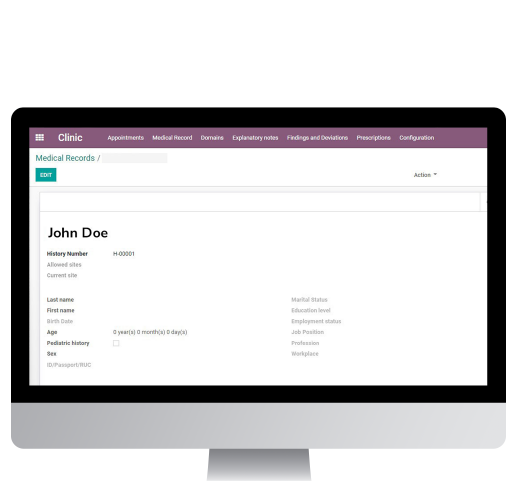
This module allows the registration of the study team’s service data from beginning to end, all the visits information, through structured data and taking into account the coordination between all the delegates; it has an information view adjusted per role, helping to improve the experience of the healthcare team, mitigating human errors, improving the information quality and reducing paperwork. It also has additional features such as:
At the Laboratory module, the site can manage the whole cycle of laboratory samples; from the request, sample collection, to the results or the shipping to external central laboratories. The results can be uploaded directly into the system by the site staff and will be reflected in the Electronic Medical Record.
Features:

Register and monitor orders for imaging studies within a research project.
Management and monitor research products, drugs and devices used within the research site; register the technical reception of products, inventory control, dispensing, return, and registration and monitoring products final destruction.
Create
Medication

Manage
Pharmacy

An easy-to-use billing and payment platform; manage your clinical trial payments per participant, visit, or activity. Generate invoices under local regulations and keep all the accountant’s records of bills to pay, suppliers, and staff expenses.
Main features:
Documents
Sign
Activities & Notes
Dashboards
Website
Website Creator
Blog
Forums
Live Chat
e-Learning
Inventory
Inventory
Procurement
Maintenance
Quality
Human Resources
Employees
Hiring
Time Off
Ratings
Referrals
Timesheet
Marketing
Marketing (social, email, SMS)
Events
Marketing automation
Surveys
User-friendly
Scalable
Web-based
Multilingual
Multisite/Multistudy
Multicurrency
Real-time collaboration
Comprehensive management of users according to role and permissions granted.
Allows the user to create a Monitor role (when remote monitoring is enabled).
Dismiss paper usage
Our Trial 360 Early Adopter Program gives a preferred rate of USD 48 per user to start with. It includes the seven characteristics of the initial core system that is enough to conduct a first clinical trial. More as follows:
The initial core system includes:
A private infrastructure with up to 100GB of storage and 5×8 functional support.
The cost of the program varies according to the size of the site, and the type and amount of training required per user.
Finance, Accounting, Expenses, Procurement, Inventory, Human Resources, Time Sheets, eLearning, Docs, etc., with a cost per application plus implementation services.
NOTE: Prices do not include taxes. Discounts between 10-20% are available when purchasing in bulk.
Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available
Around 70% of our clinical research experts are Latin American professionals with global experience
Schedule a meeting with us!

Integra IT.
For Sites.
Other Solutions.
For CROs.
About us.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |