Integra IT provides customized software development services in response to each client’s necessities and challenges in healthcare and clinical research. Our dedicated development team allows us to create, set up, and launch robust and customized tools in record time.
We have comprehensive experience in data management and processing at a country scale, successfully operating nationwide and multicountry-wide support.

Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available
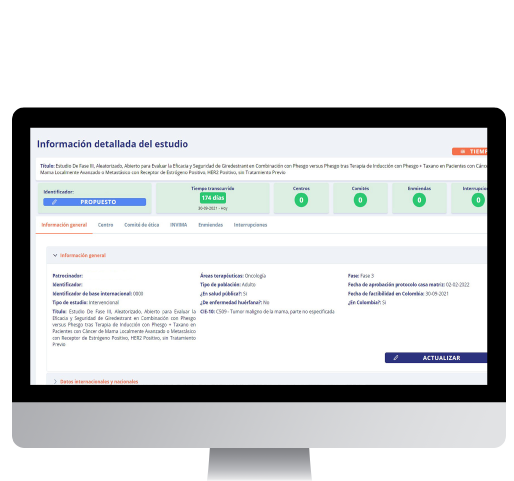
The Metrics software is a tailor-made tool that enables the user to record and control clinical trial development for countries, local authorities, and associations, aiming for time management in a unified database. Check trial status, establish key indicators for effective decision-making and optimize resources and efforts strategically to make the local clinical trials field more competitive and efficient.
Currently, the Metrics solution is being used by the most important pharmaceutical, I&D, and research organization in Colombia (AFIDRO), contributing to clinical research development and improving tracking trial progress. Likewise, having this kind of tool helps to attract investment and find new ways to promote clinical research initiatives in middle and emerging markets.
Schedule a meeting with us!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |