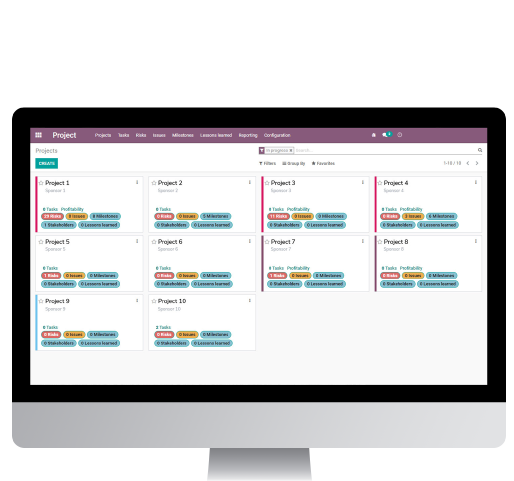
CTMS 360 is a GCP-compliant web-based, easy-to-use platform developed so CROs can speed up processes and improves quality across the clinical trial project management discipline. This solution provides project management tools like Gantt, task views, lists, and a database for sponsors, sites, and countries. The system also tracks risk monitoring, deviations and violations, milestones, stakeholders, and outcomes.
Our CTMS 360 is an all-in-one project management application that can be integrated with billing and human resources modules.







Keep a record of issues in the project operations, allocate category, impact, causes, preventive and corrective actions, and the person in charge.
Manage, track, and register the project stages. For example, research site activations, contracts, or closings.
All features include an audit trail.
Discuss
Calendar
Contacts
Admin Generic Group
Website
User Sessions
Review
Inventory
Payroll
Timesheets
Surveys
Collaborators
Attendances
Time Off
Expenses
Approvals
Employee Books
Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available
Schedule a meeting with us!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |