TrialPal is our eCOA (electronic Clinical Outcome Assessment), ePRO (electronic Patient Reported Outcomes) and Site Mobile and web application used in vaccine clinical trials and decentralized clinical trials.


TrialPal is our eCOA (electronic Clinical Outcome Assessment), ePRO (electronic Patient Reported Outcomes) and Site Mobile and web application used in vaccine clinical trials and decentralized clinical trials.
Setup Study Protocols in minutes and prepare Screenshots for IRB Submission in a Day.
Subject’s easy-to-report, friendly Web, PWA, and Mobile App with offline functionality.
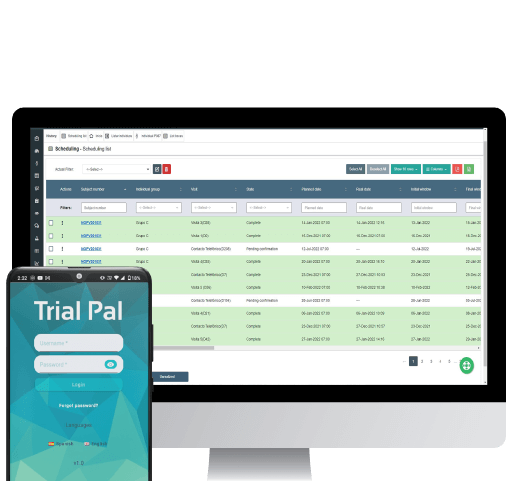
The Site’s management console, with reports, eDiary view, eDiary Assessment, and subject follow-up.
TrialPal is an intuitive and easy-to-use tool that monitors clinical trial participants and the study, through dashboards combined with a user-friendly interface created to generate data directly from the trial participants






Our TrialPal dashboards provide an overview of the trial status. This is accomplished with on-demand indicators such as; symptomatology, intensity per participant by group or site, as well as reports and visit completion.
Click to see full image
The dashboards allow detailed tracking of AE and SAE, and other key variables to make data-driven decisions throughout the study. The practical design makes it easier to do analytics and presentations.
Click to see full image

Create any eDiary and any other trial-specific forms in as little as 1-2 hours as per protocol specifications.
With our conciliation module you can review and conciliate easily all the symptoms reported.
This allows users to activate additional modules such as Chat, eConsent, and Telehealth (Beta version), it also provides a way to enable hybrid or Decentralized Clinical Trials (DCT) when integrating TrialPal with Trial360 and EDC.
To enhance interoperability between site CTMS, EDCs, or any third-party application.
In TrialPal you can made forms for your surveillance reports using a design mode; with drag-and-drop features, and all the functionalities you need to create the perfect form for your study needs.
TrialPal App works in offline mode. The App saves the report on the device, and once it is back online, all the pending data is sent automatically.
Besides, with the traveler mode, you can go anywhere worldwide and keep reporting the days you are out of your time zone.
Easily set up vaccine clinical trial eDiary or surveillance forms for symptom monitoring, including: subforms, grading, alerts, and AEs – SAEs real-time notifications to stakeholders, where sites can also act quickly upon receiving subject reports.
With the Trial Site App, physicians can manage reconciliations, and with Trial API, data is sent automatically to the eCRF or EDC system.
Reliable
Configurable
Real-time information
User-friendly
Traceability
Works in Mobile and Web devices
We studied the performance of a smartphone application-based participatory syndromic surveillance (PSS) system for collecting syndromic data (acute febrile illness [AFI] and acute gastroenteritis [AGE]) to detect dengue virus (DENV) and norovirus (NoV) on a cohort of children living in a low-resource and rural area of Guatemala.
Read the article “Performance of a Smartphone Application-Based Participatory Syndromic Surveillance System for Acute Febrile Illness and Acute Gastroenteritis in Rural Guatemala“ by D. Olson, Takeda Pharmaceuticals: Consultant, Grant recipient. R. Zambrano, Integra IT: Employee and Shareholder, Salary. S. Rodriguez-Castro, Integra IT: Employee and Shareholder, Salary. E. J. Asturias, Takeda Pharmaceuticals: Consultant and Grant Investigator, Grant recipient, at Oxford Academic.
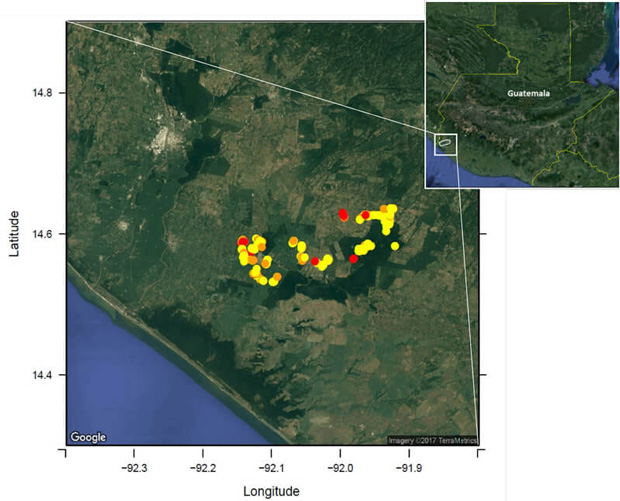
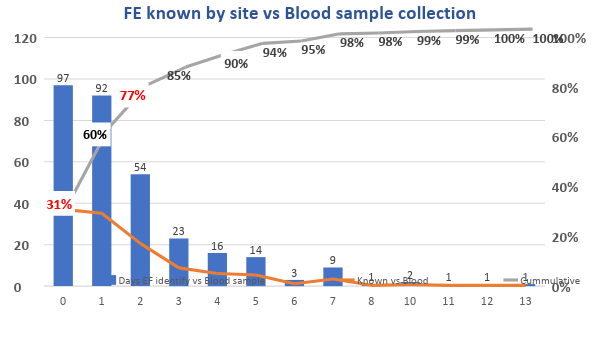
FE known by site vs Blood sample collection
Clinical Trial: 935 subjects
Phase 2 Dengue Vaccine
Site: Cevaxin Panama
Schedule a meeting with us!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |