
Importance of Pentesting in Software used in Clinical Studies
Cybersecurity: Critical in Clínical Trials In recent years, the number of cyberattacks has increased due to the proliferation of viruses, malware, and new and
The combination of three of our best solutions is made to be an easy-to-use platform for your site’s needs.
Integrating our EDC, eCOA/ePRO, and trial management system is easy and seamless.
Monitoring study participants and the study in general, minimizing processes, reducing costs, keeping track of diaries and registers, and monitoring the clinical trial data; all in one integration.
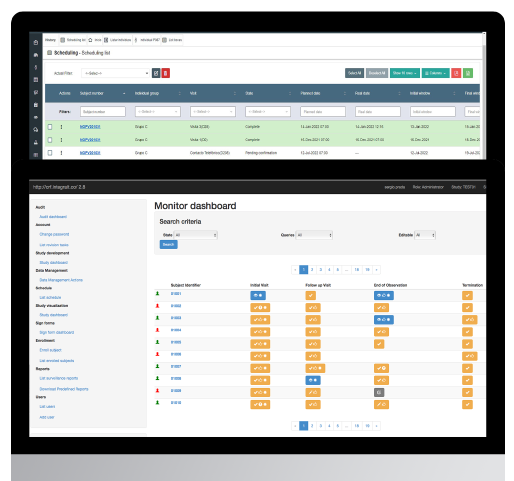
For Integra IT, interoperability is a premise; we deliver fast and impeccable integrations with third-party systems like RAVE.

Below you will find an integration example. Our solutions include an easy-to-setup API that extracts data from our ePRO or eCOA to Site CTMS and, along with our eCRF Importer, is then transferred to our proprietary eCRF or a third-party study EDC system like RAVE. In the RAVE example, we use Rave Web Services to make both systems communicate with each other. We have also developed an integration checker that review and compares data between systems, so the research site can be confident that all data was transferred successfully and that data integrity is being met.
Notes:
Integrations take 2-3 weeks depending (approx) on study forms (# variables, visits, e-Dairy days).
Testing and validation take one week (approx) to make sure integration works flawlessly.
Rave dedicated user needed to authenticate transactions via RWS.
Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available
Around 70% of our clinical research experts are Latin American professionals with global experience

Cybersecurity: Critical in Clínical Trials In recent years, the number of cyberattacks has increased due to the proliferation of viruses, malware, and new and

Computer System Validation is a premise for Integra IT to ensure that data and solutions are reliable; validating all of our solutions is one of

Integra IT understands and has the solution to an issue commonly faced by healthcare systems and clinical trials around the world: interoperability. Seamless integration between
Schedule a meeting with us!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |