The Study Tracking System (STS) is designed for companies that need to manage clinical trial monitoring visits reports and related information aligned with industry requirements such as CROs.
STS is a web-based application designed to create, review, and approve the monitoring visits reports developed during the conduction of research studies. It includes scheduling monitoring visits and access to the study database, where action items, protocol deviations, safety information, SDV logs, monitoring visits reports submission/approval timelines can be consulted, and reports obtained to follow the trial status closely. This information can also be analyzed through customized dashboards for a deep real-time analysis of the trial metrics and relevant information, allowing timely decision-making.

Multi-study
and multi-site

Reports review and status

Download the reports in PDF format

Complete visits forms directly into the system

Complete data entry with compulsory fields settings

Assign projects per role

Audit feature for audits and processes validation

Import mass data from Excel

In the STS, CRAs (Clinical Research Associates) complete their monitoring reports without losing information. They can save the data in real-time, plan monitoring activities, and consults the trial/site information using the research study database.
With STS, PMs (Project Managers) can handle multiple sites, review reports, and manage trial information. Additional staff can access information to support planning and follow-up activities (e.g., CTAs) and perform oversight activities (e.g., Line managers, Quality Area). Within the STS, Monitors and Project Managers certify that all processes are carried out according to best practices by signing off tasks electronically.
As per trial requirements, the CRO can set up:
This module overviews each visit’s current status (created, pending approval, or approved).
The Reports creation: This module allows the CRA to collate information during the visit, and complete the site visit report with the information gathered at a later date. Customize sections like site training, study samples, facilities and equipment, Serious Adverse Events (SAE), general comments, informed consent, and Action Items.
For example, in the Action Items section, the CRA can register protocol deviations, create issues, assign open and due dates, determine the actions required, and add notes.
Create
Report
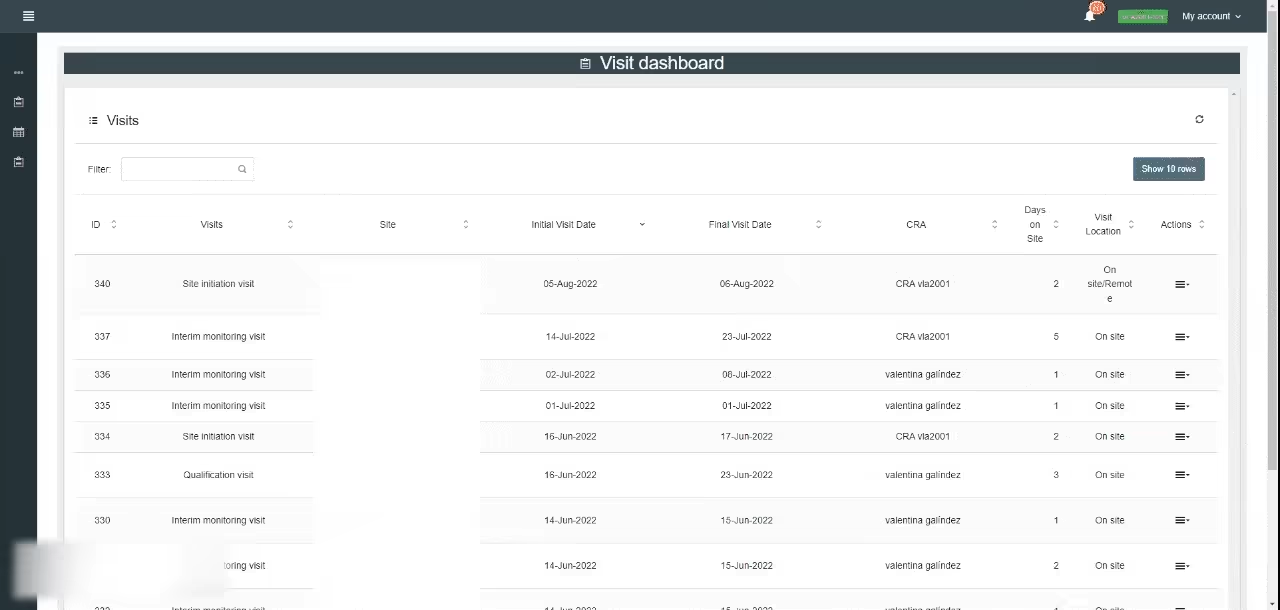
Report
Dashboard
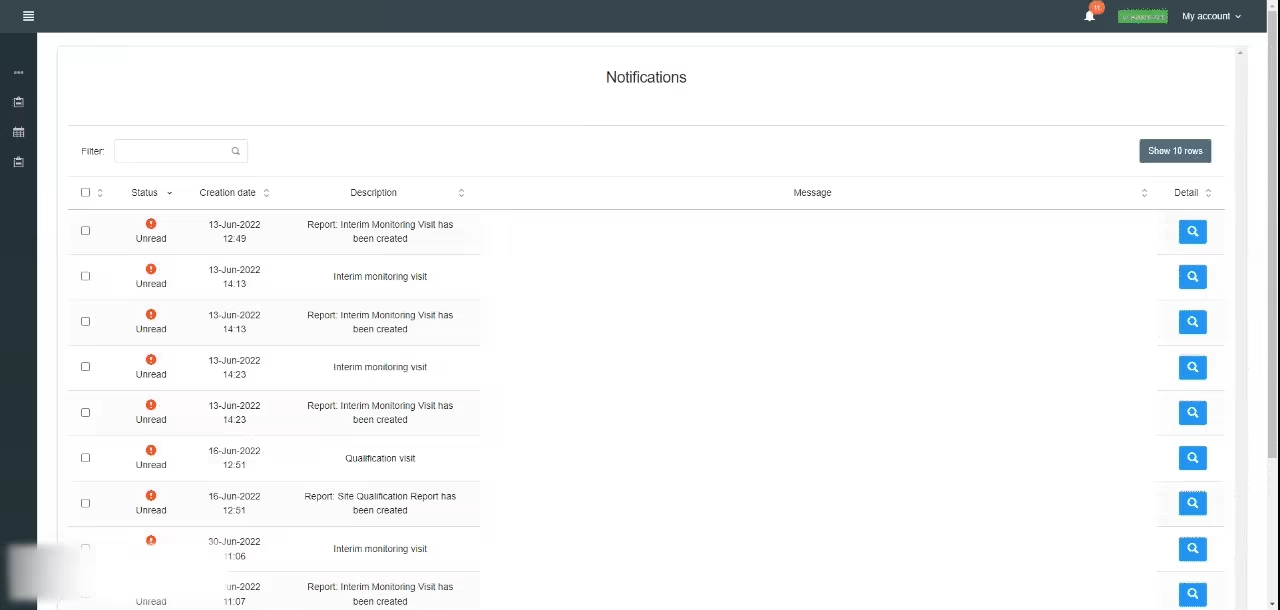
Access dashboard panels with personalized metrics per study and get an overview of indicators like SAE reports SAE times, report type, and record dates of sponsor notifications in a graphic view.
This functionality is a time-saving tool for CROs and enables them to spend less time on transcription and paper tasks, so they can focus on analytics and efficiency.
Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available
Schedule a meeting with us!

Integra IT.
For Sites.
Other Solutions.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |