
What is Clinical Data Management?
Clinical trials are essential to advancing medical science and providing patients with novel treatments. The optimization of clinical trial operations depends critically on effective data
Intega IT, Viedoc Partner for Latin America and Emerging Markets
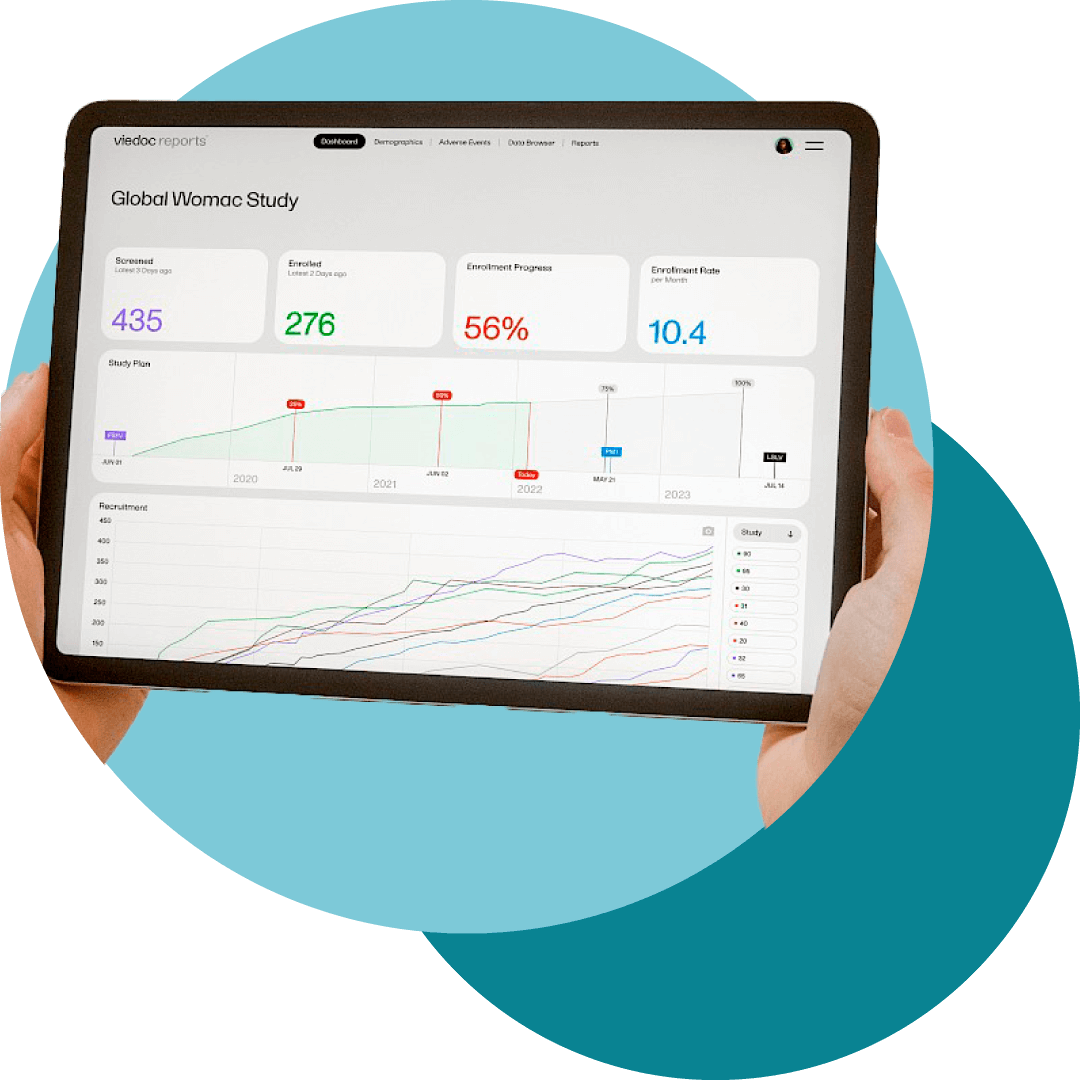

Intega IT, Viedoc Partner for Latin America and Emerging Markets

This package provides a cost-effective and user-friendly electronic Case Report Form (eCRF) and web solution. Paired with our TriaPal ePRO and eCOA mobile solutions, we enhance clinical data management through superior deliverables, timely execution, and exceptional quality.
Our approach emphasizes high efficiency, ensuring transparency, data integrity, and data quality throughout the clinical trial process.

To know more about Viedoc
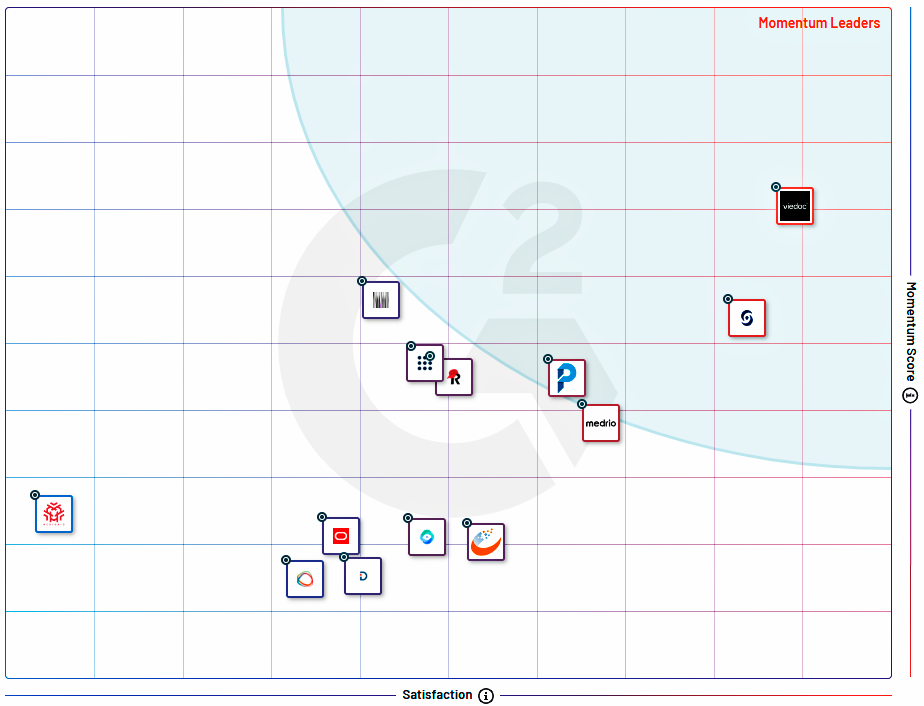
Affordable (accessible) for small and medium size Sites, CROs, and Sponsors
Real-time data for data-driven decisions
Faster start-up studies
Interoperability (integrations between own and third-party systems)
Validated and GCP-compliant
On-demand scalable architecture (per solutions, modules, and features)
Expertise in low and middle-income countries
Vaccine-exclusive features available

Clinical trials are essential to advancing medical science and providing patients with novel treatments. The optimization of clinical trial operations depends critically on effective data

As the demand for efficient and streamlined data management in clinical trials continues to grow, with numerous vendors offering a wide range of features and

Integra IT has developed reliable eCRF software solution and Data management services priced to fit your budget and excels in delivery, quality, support, and time.
Schedule a meeting with us!
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |