Sites can get advantages of tools like structured EMR data to provide standard and consistent data entry in specific fields to conduct research (1) and can be used for surveillance and monitoring. A comprehensive software platform designed for sites can make them achieve a ROI (Return on Investment) by impacting multiple activities regarding the operations and, for example, reducing trial days costs, improving the enrollment rates, and the study launch dates.
Here we summarize the main advantages sites can get by acquiring a compliant software solution:
Adherence rates: Increasing participant interactions, participation, and engagement while mitigating site burden can be enabled by technology adoption. With automation, sites can have accurate visit follow-ups, avoid visits out of the window, and get true-time study performance indicators. When a site’s operation turns digital, more of its efforts can focus on improving the patient experience and ensuring as few protocol deviations as possible.
All-in-one: A common site concern is the centralization of its activities. For a proper digital transition, sites may look for an all-in-one modular architecture solution with a user-friendly interface and cloud-based infrastructure that adapts to the site size. Likewise, it is desirable to work with a local IT support team in the local language and check the supplier’s technical, operative, and cybersecurity processes, pentesting, and CSV.
Real-time data and visibility: Gaining visibility during the trial is a precious virtue a software solution can offer. A real-time overview of the trial status can alert on delays, speed up processes and help to make decisions over the site’s resource allocation and overall each trial. One of the main challenges sites face is time, and having a clear view of key performance indicators to present to the sponsors leads to significant advantages. For example, while conducting Investigator Initiated Studies (IIS), sites can gain early outcomes and control capacity with a CTMS, automated executive dashboards, and smart scheduling.
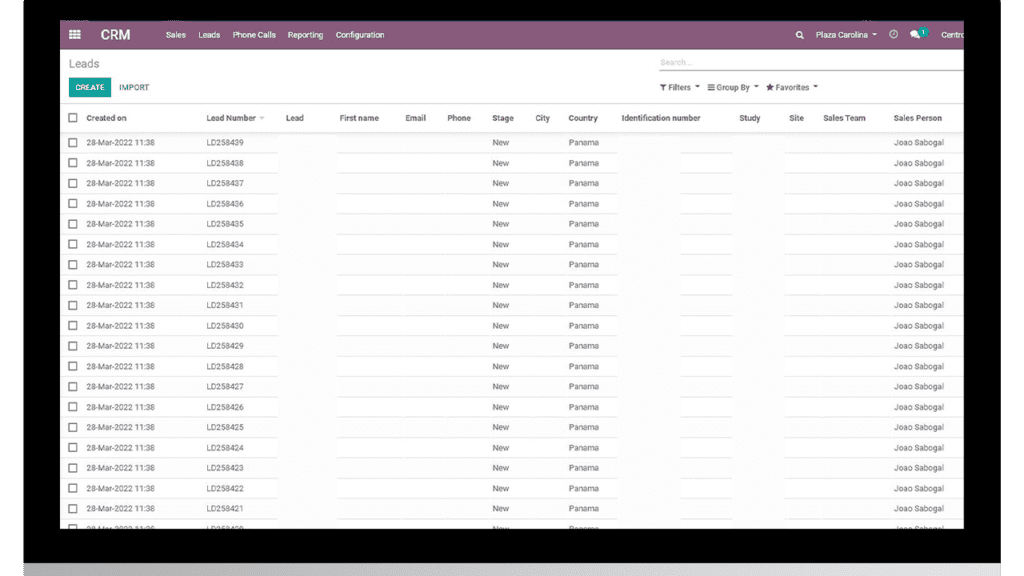
Workflows: Streamlining clinical trial procedures in sites is very useful to have a clear research subject flow and to achieve as few protocol deviations as possible. Compared to spreadsheets, a technology-based tool encourages collaboration within the study team and keeps operations standardized, guaranteeing quality controls with features like audit trails. For example, systems like Trial360 allow to control and track the entire Lab samples cycle, from the take until the results are recorded in the EMR.
Compliance: A common pain in site operations is compliance. But now, through experienced vendors, sites can guarantee compliance with multiple validation guidelines and standards like FDA 21 CFR Part 11, ICH-GCP, EU Annex 11, HIPAA, and ALCOA. These industry guidelines facilitate using features like e-signatures, audit trails, data security with user roles, and the system’s general quality.
Hopefully, these 2023 sites will still join the digital transformation and continue benefiting from software technology. Thinking of early adopters and low and middle-income countries sites, Integra IT designed Trial360 as a complete, cost-convenient, and high-quality solution for sites. Thanks to its complementary modules and scalable architecture, it can grow on the go with the site with a pay-as-you-go model.
Join our Trial360 Early Adopter Program and let us guide your journey to a modern and efficient site.
References:
- Manca D. P. (2015). Do electronic medical records improve quality of care? Yes. Canadian family physician Medecin de famille canadien, 61(10), 846–851. Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4607324/




