Integra IT software solutions has been essential for developing the Fiocruz vaccine Phase 3 trial for SARS-CoV-2 in Brazil, achieving efficiency in data collection and organization. This project collected information from 10,000 volunteers for a year, managing their data via Integra IT’s TrialPal and PFS tools.
The project was carried out in Brazil, a Latin American nation with 211 million inhabitants. Its geographic extension and population density allow the researchers to draw conclusions based on crossing variables derived from climate, regional conditions, and common diseases.
This trial had participants from five states: Bahia, Sao Paulo, Rio de Janeiro, Rio Grande do Sul, and Rio Grande do Norte, all bordering the Atlantic Ocean; each of these territories had one or two sites in charge of the clinical research operation and the participants coordination. The project was an initiative of the Oswaldo Cruz Foundation (Fiocruz), which have more than 70 years of experience in the vaccine production field, and the University of Oxford.
For the storing, harnessing, and processing of the information of the 10,000 participants for 12 months, it was pivotal to use technological tools to enable participants to provide details in a simple way. Due to the pandemic, it was essential to have real-time access to symptomatology reports and alert the sites to make a timely detection of COVID-19 cases.
Additionally, those in charge of receiving the data had control over how frequently the sponsor was updated on trial progress and could adapt data to meet trial desired outcomes.
Integra IT Technology
applied to the Fiocruz Project.
Integra IT, with its TrialPal and Patient Follow-up System (PFS) tools, ensured that the process of delivering and processing information was secure and accurate for the stakeholders. We implemented rigorous security and data management practices to keep data protected, Furthermore, Integra IT’s simple processes allow for efficient sharing with clinical trial stakeholders.
The data collection tools allowed the optimization of processes and the detection of Covid-19 cases almost in real time. Tasks that usually could take up to three days, in the Fiocruz Project only required hours, like the set-up of the protocol in the system and scheduling visits, thanks to the advanced and easily configurable technology offered by Integra IT.
Another way to improve the execution of a vaccine trial was to take advantage of the raw data by converting it into usable decision support dashboards easily customizable with different kinds of filters, transforming data into information, and giving the sponsors fast decision support capabilities in their study. For example, in this study, the sponsor was able to identify the social and demographic variables of the population with more COVID-19 cases detected and then make closer surveillance of their risk conditions.
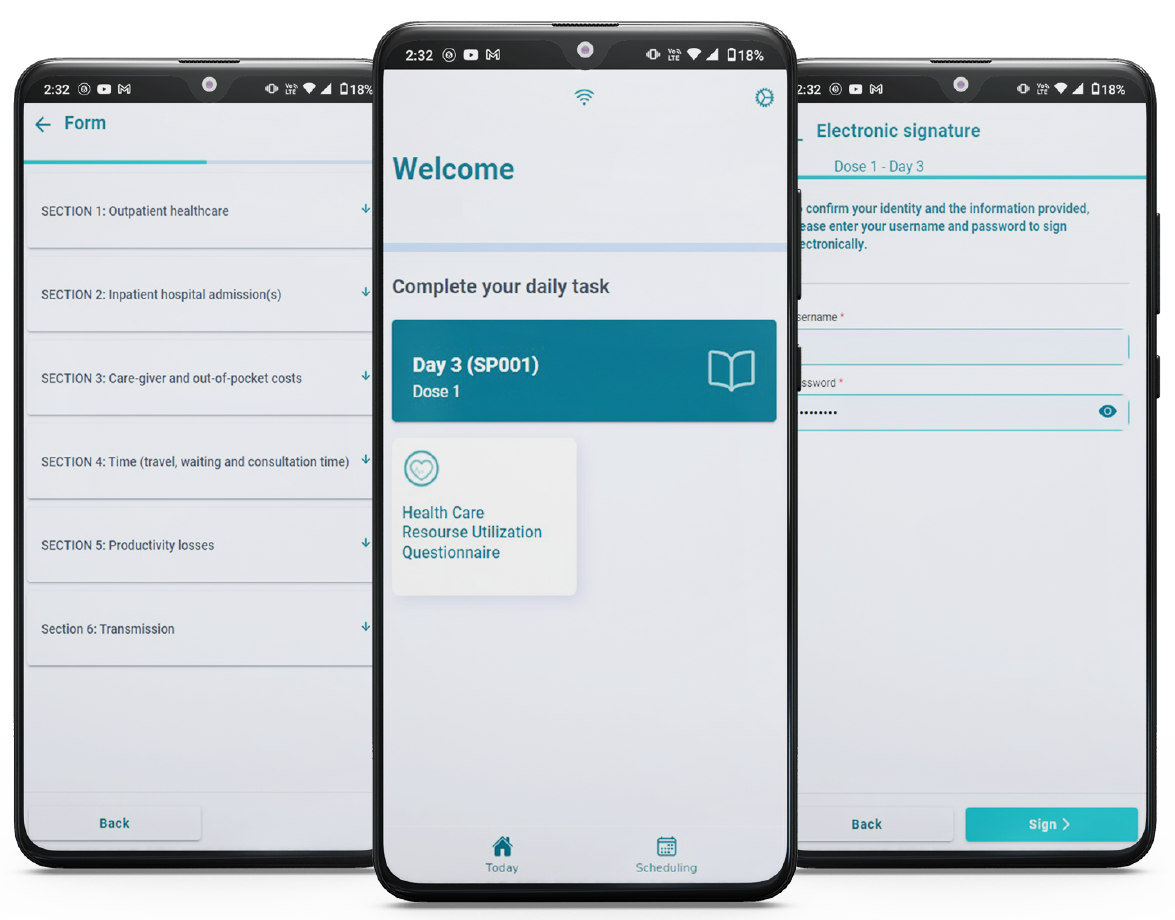
In Image 1 we can see an example of how participants provide the información in a user-friendly interface in the eDiary module. Color codes, notifications, and logic flows help the participant or the parent, in the case of minors, to report symptoms after each visit and dose.

Figure 2 shows an example of the scheduling module of the PFS, which allowed the creation of the visits in the system, in order to track the status per group, stage, planned date, real date, initial window, and the final window, and if the visit was a phone call or in person. With this interface it was easier for the researchers to review the status of the scheduled visits and export the data in different formats.
Additionally, when the information arrived, the researchers were able to break down and catch early insights and compare the preliminary results. For example, they could identify if there were adverse events (AE) in the seven days after the first shot of the COVID-19 vaccine.
What is TrialPal?
TrialPal is a mobile surveillance application where the trial participants record symptoms or any new developments of possible serious adverse events (SAE) and adverse events (AE). The app has a user-friendly interface that improves the participant experience and is designed to be intuitive and inclusive for every age user.
- According to 99% of people who have used it, it is easy to use.
- Enables the instant gathering of information.
- Available on Android and IOS
What is PFS?
It is a web-based application that centralizes all the information gathered by the mobile applications, including TrialPal, allowing trial conduct to be operated by recruiting sites. This includes scheduling participant visits, monitoring report completion by participants, and easy data export.
- Works for desktop or mobile
- Reduces errors and improves efficiency
- Reduces the workload of reporting information requested by the sponsors.
- Has a variety of modules such as scheduling, households, and lab sample management.
A Success Study Case about Efficiency
A large team based in Sao Paulo was in charge of the analysis of the Project; they were responsible for attending to the entire flow of subjects participating in the trial. However, during certain months, the presence of all the collaborators of the site team was not necessary. Thanks to the PFS tool, it was possible to detect when more or fewer staff were required based on the participation flow of the project collaborators. Thus, they could plan the demand, and balance workloads to serve the participants better without delays. See the graph below:
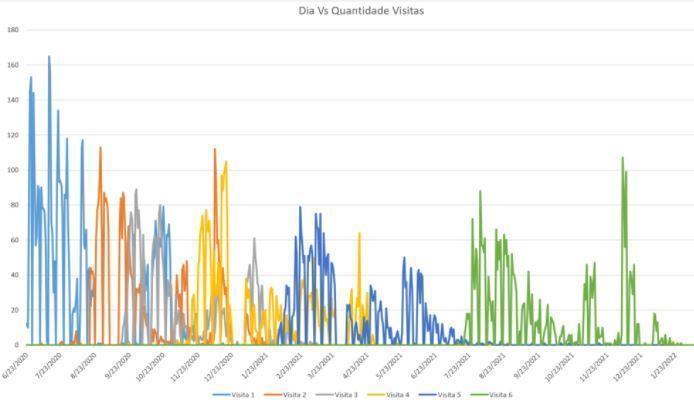
Graph 1. Visits per day
As a result, based on the information collected, this graph was created, which shows the number of visits compared to the day and identifies the moments when more or less logistical support was needed in the site.
Increase your adherence reporting rate in any vaccine trial working with TrialPal, learn more:




