Clinical research protocols are carried out following the International Council for Harmonization (ICH) guidance on Good Clinical Practice (GCP), which outlines the requirements for corrective and preventive actions to be identified, reviewed, reported, and taken appropriately to ensure patient safety and maintain data integrity.
What are deviations from the protocol?
According to ICH E3 guidelines, protocol deviations are any changes, divergences, or deviations from the study design or procedures defined in the protocol.
How can deviations affect a protocol?
Deviations from the protocol may affect the quality and integrity of the data, the rights, safety, and well-being of participants, and the scientific validity and reliability of the study data.
How do we prevent deviations from the protocol?
Some strategies that help improve data integrity and protect study participants are:
- Electronic data capture
- Training the site team in the therapy area and protocol.
- Regular and effective risk-based monitoring, focusing on critical data and processes
- Have a quality management system and standardized operating procedures.
- Statistical approach in the review of the accumulated data of the study.
- Supervision by the ethics committee
Key features of Findings and Deviations Management in Trial 360
- Centralized Management: Trial360 integrates all stages of the study, and the detection to the resolution of findings and deviations, in an intuitive and easy-to-use interface.
- Real-Time Tracking: Maintain constant control over each finding and deviation through real-time updates, allowing for a quick and efficient response.
- Custom Reports: Generate detailed and customizable reports to analyze trends, identify areas for improvement, and maintain transparency at all times.
How is the handling of findings and deviations in Trial 360?
In Trial360, we have the findings and deviations module, in which a user with permissions can create a finding, record the type of finding, the person responsible, category, cause, corrective and preventive actions, criticality, related subjects, report to the ethics committee (if it is a deviation), it is possible to relate the visits, adverse events, laboratory studies or quality reviews that may occur.
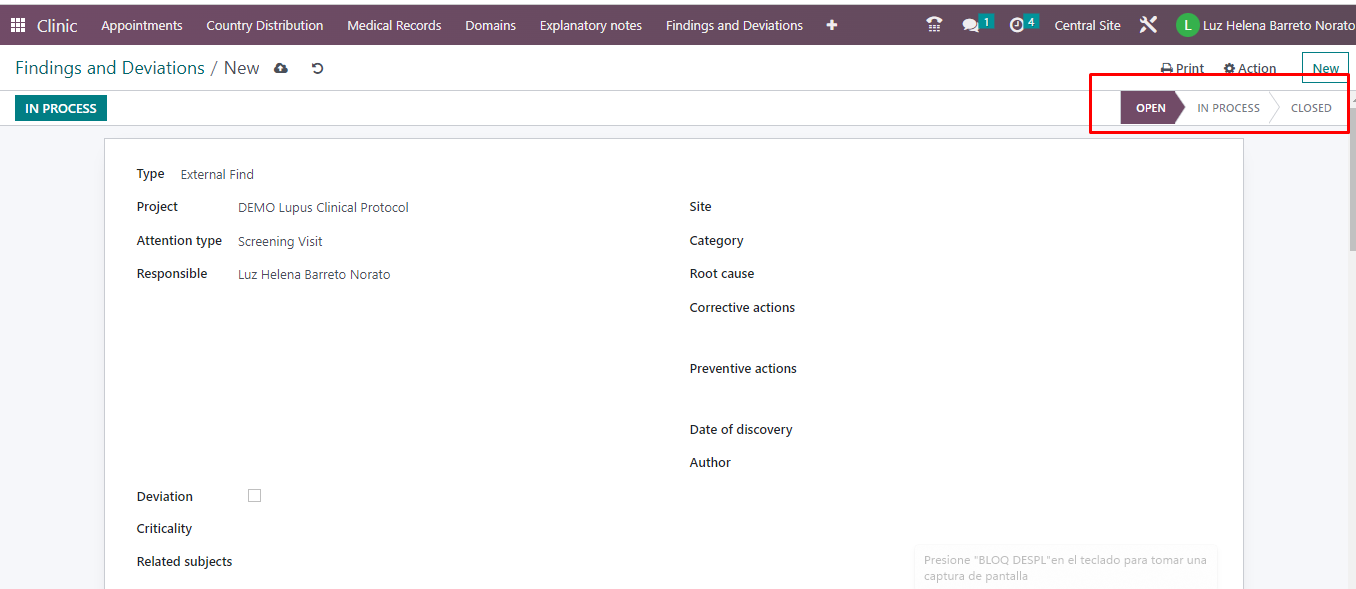
- The finding or deviation is tracked through states: open, in process, closed.
- Monitor roles can create findings and deviations. However, the monitor role will only be able to view the records created by itself.
- It is possible to plan activities associated with a finding that is notified via email and through the activities icon, which is visible every time a user enters Trial 360.
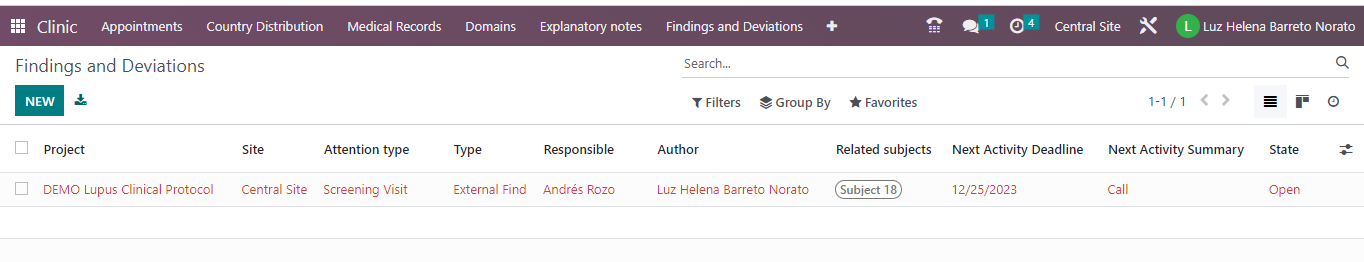
- In the general list of findings and deviations, the records are displayed with a traffic light that indicates whether they have activities pending execution or with a delay date.
Despite increased attention to monitoring, audits, and regulatory inspections, managing protocol deviations still needs to be improved when conducting clinical trials. It is of utmost importance to have systems or tools that allow the complete management of a research protocol, including the management of findings and deviations.
Questions about findings and deviations management in Trial 360?
References
Chodankar D. Impact of protocol deviations on the clinical study. Perspect Clin Res. 2023 Apr-Jun;14(2):47-48. doi: 10.4103/picr.picr_69_23. Epub 2023 Apr 13. PMID: 37325573; PMCID: PMC10267991.
Protocol Deviation Process Guide. TransCelerate. 10 Aug 2020. https://www.transceleratebiopharmainc.com/wp-content/uploads/2021/03/Protocol-Deviations_Process-Guide_March-2021.pdf
Attachment C: Recommendation on Protocol Deviations. the U.S. Department of Health and Human Services (HHS). https://www.hhs.gov/ohrp/sachrp-committee/recommendations/2012-march-30-letter-attachment-c/index.html




