Electronic Clinical Outcome Assessments (eCOA) and Electronic Patient Reported Outcomes (ePRO) are two types of electronic data collection methods. While they may seem similar, there are some differences between them that are important to acknowledge.
ePRO involves patients reporting their own health outcomes directly to their healthcare provider or research team. This is usually done through online questionnaires via the web or Mobile applications. An ePRO tool is designed to capture patient-reported data in a standardized and consistent way and is often used to assess symptoms, quality of life, and other patient-reported outcomes. (See: TrialPal, the Participant’s best Friend)
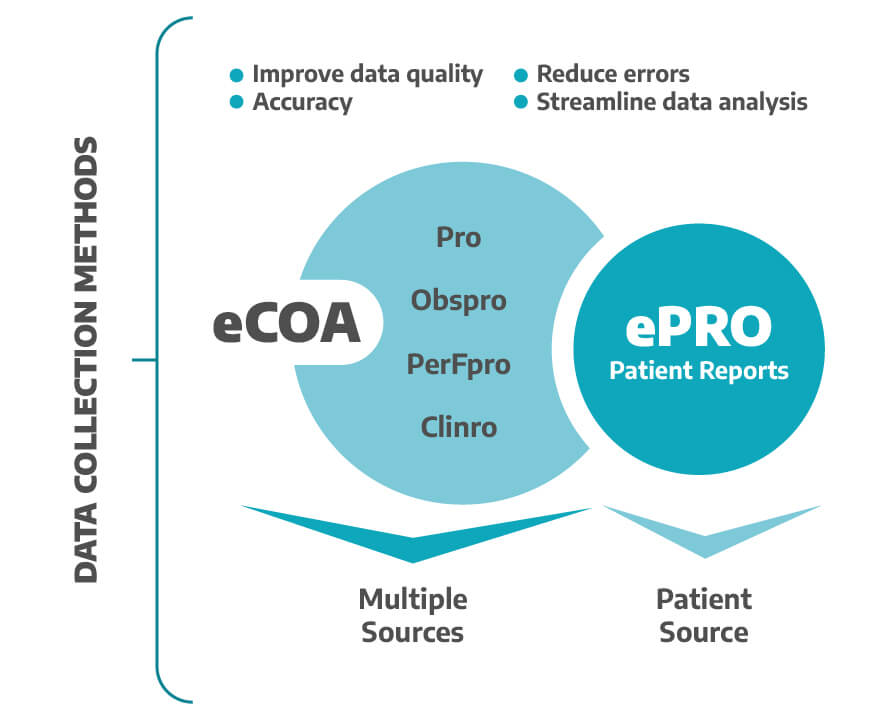
An eCOA is a broader term that includes different kinds of electronic data collection methods used to collect clinical trial data, including ePROs.
Assessment of a clinical outcome can be made through a report by a clinician, a patient, a non-clinician observer, or through a performance-based assessment. Types of COAs include Patient-reported outcome (PRO) measures, Clinician-reported outcome (ClinRO) measures, Observer-reported outcome (ObsRO) measures, and Performance outcome (PerfO) measures (1).
Another key difference is the way the data is collected; ePRO is typically self-reported by patients, often through mobile Apps. eCOA, on the other hand, can involve a variety of data collection methods, including clinician assessments, performance tests, and other types of data capture.
One of the main differences the source of the information; while the ePRO is focused specifically on patient reports, the eCOA can include data from a variety of sources, including healthcare providers, study staff, and observers.
They also share common benefits, ePRO and eCOA are both methods that help improve data quality and accuracy, reduce the risk of data entry errors, and streamline data collection and analysis. Additionally, both can help improve patient engagement and satisfaction.
In conclusion, while ePRO and eCOA are similar in some ways, they are also different in terms of the types of data being collected. Understanding these differences can help choose the right data collection method for your clinical trial’s specific needs and goals.
Also it is important to consider how sites and the study team are informed regarding specifics of the reported outcomes so they act quickly when data is reported. Real-time data available is needed to improve the decision-making process.
Contact us to learn more about eCOA and ePRO tools and how combined with the right set of services can produce enormous value at the different stages of a clinical trial, like safety and efficacy for a vaccine clinical trial.
References:
- BEST (Biomarkers, EndpointS, and other Tools) Resource. http://www.ncbi.nlm.nih.gov/books/NBK338448. Accessed 12 Oct 2021











