Around this season, many research sites are planning and projecting their budget for the upcoming year 2024; this is an excellent opportunity to consider a key investment in technology and data-driven management that will bring efficiencies and savings in the short, medium, and long term.
By implementing Trial360, sites can go paperless, as well as reducing the amount of licenses and software they use, centralizing all clinical trial conduct activities (CTMS, HME/eSource, Pharmacy, Laboratory), as well as all administrative tasks (Human Resources, Finance, Purchasing, Marketing. etc.), into a single robust and validated system (FDA 21 CFR Part 11, ICH GCP, local regulations).
In addition, Trial360 has a flexible modular system for progressive or total adoption, with prices adapted to sites of any size. Thus, you can turn this:
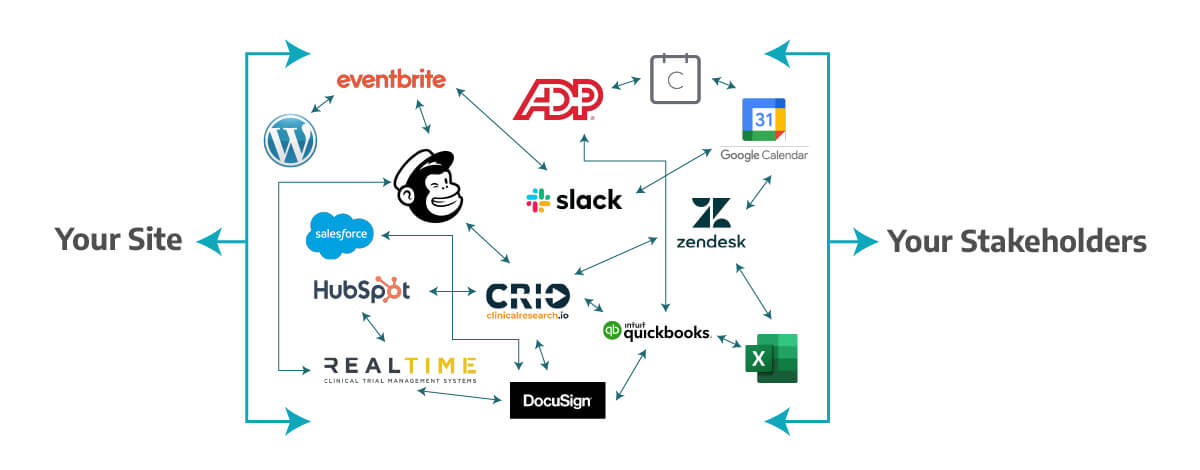
Into This:
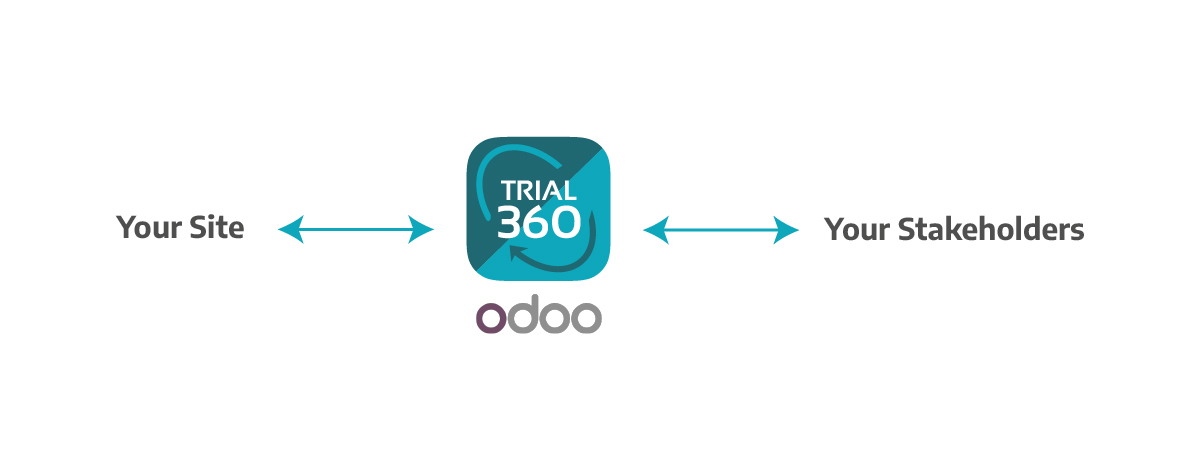
We are available to support your technology evaluation, proposal submission, and change management process. Include Trial360 in your 2024 budget, and build your next budgets in the same system, with centralized, real-time information and comprehensive management indicators for all processes.




