Peru, a country of nearly 33 million people, was hit by the COVID-19 coronavirus pandemic mainly because the health system, like that of many other countries, was not prepared for a health challenge of such a magnitude.
According to official figures, there were only 100 intensive care unit beds and 300 hospital beds in the whole country at the start of the pandemic.
As a measure to curb the spread of this new coronavirus, one of the strictest quarantines in the region was decreed, which had a considerable impact on the economic situation.
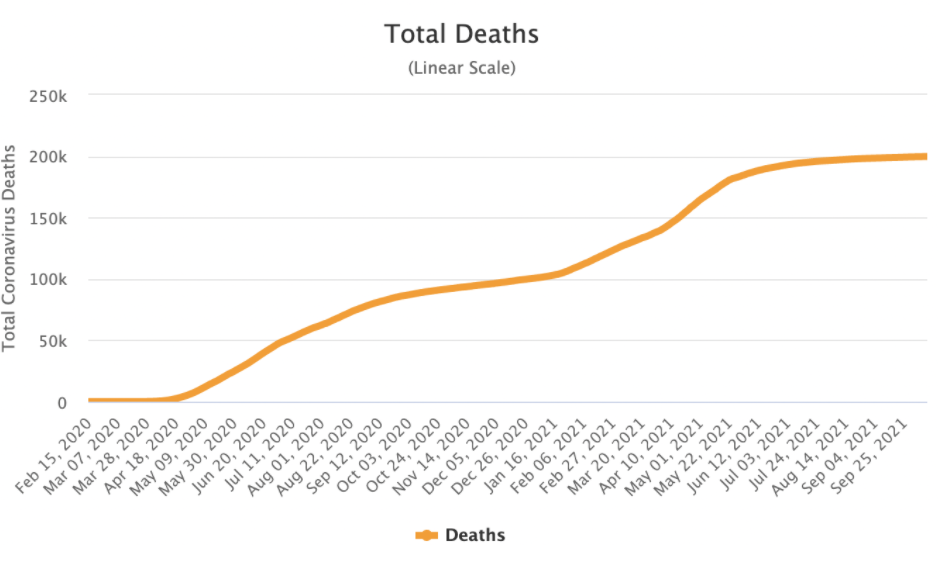
But the economic losses did not compare with the human losses. Peru was one of the countries with the highest number of deaths per month due to COVID-19. By October 2020, deaths had already reached 100,000 and no commercial vaccine was in sight in the short term.

The sponsor Curevac and the CRO PRA conducted a Phase 2 dose confirmation clinical trial to evaluate the safety, reactogenicity and immunogenicity of the COVID-19 CVnCoV vaccine. Peru enrolled 335 participants over a 3-month period.
The information gathered from this group of volunteers was collected using three tools developed by Integra IT:

The challenges for
Integra IT in this study
Each study brings with it a different scenario. This study evidenced the cultural barriers in the adoption of technology, the need to provide more than just technology, a set of support services and training to learn and respond to unforeseen circumstances.
When Integra IT took over this project, it became evident that one of the main challenges was that the project’s lead study site was working manually on paper and some spreadsheets. In fact, the field workers did not have computers.
In addition to not having the technological support, their training was not focused on transferring the information to the digital field and the data collected was recorded on spreadsheets that were completed on a daily basis.
Integra IT assigned a focal point for on-site accompaniment and support with the objective of instructing and supporting participants in the use of the mobile application on their cell phones, as well as the delivery of pre-configured cell phones with data plans for those who did not have a device.
The site staff was not trained to help the study participants in case of any inconvenience, as many of them also did not know how to use the application despite the training provided. Therefore, Integra IT took on the role of identifying, responding to and resolving any issues regarding the use of the mobile applications, as well as the use of the web application by site staff.
Integra IT saw the need to train the staff in charge of collecting the information to avoid scenarios such as the accumulation of tasks and create a database of knowledge and provide accompaniment to the health professionals who now had to use the application.
The good practices with staff members and their projection towards the participants led to positive conclusions such as those described below.
The Integra IT PFS comes with modules tailored to the customer needs. For more information
Results
and learnings
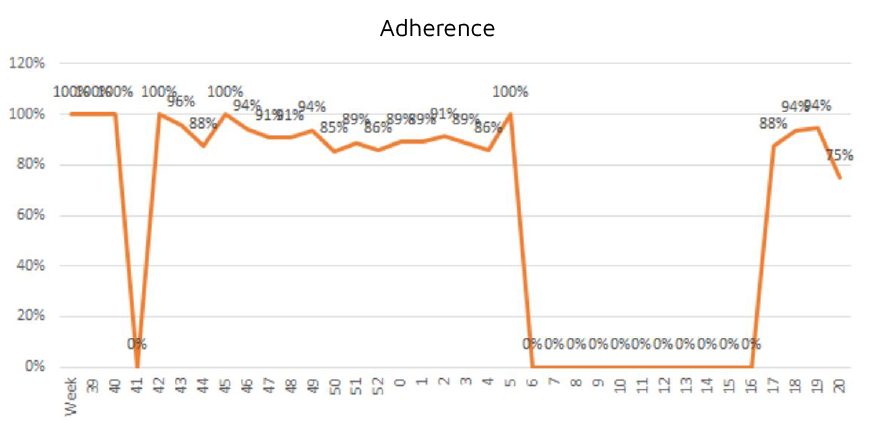
In terms of results, the most important aspect for Integra IT was the positive effect in relation to the applications developed. The adherence achieved with the participants was very good, with a daily average of 90%. This allowed us to support the objective of identifying Covid cases within hours.
The conclusion is positive considering that it was the first time that the study site used this type of technology and that while the study was being conducted, a process of organizational transformation from manual to digital was implemented, which, despite some setbacks, went ahead in a very satisfactory manner.
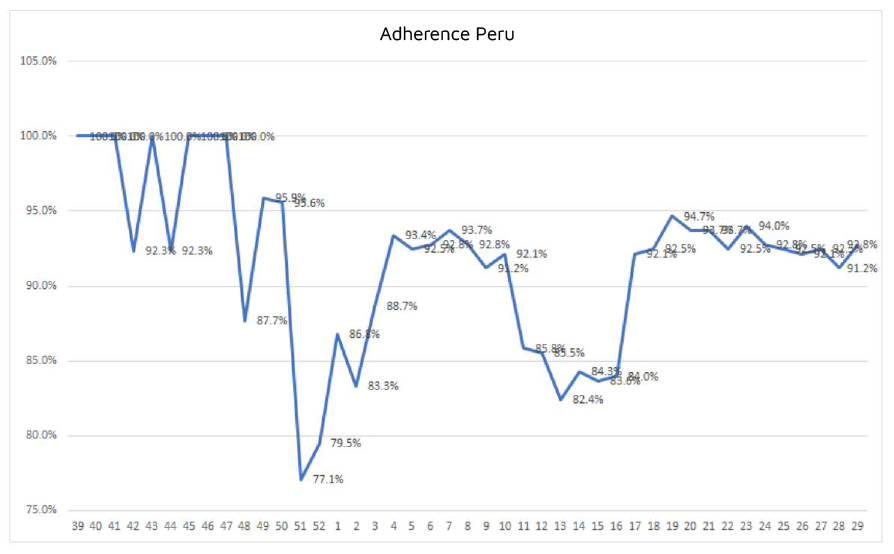
Regarding the Vigilant-e module for surveillance and participant reports, it can be concluded that adherence was around 92%. Although there were times when the percentage dropped to 77% at its lowest point due to national holidays or vacations, it was possible to rapidly increase use thanks to constant reminders and re-training, together with the site’s willingness to call those who had not reported.
The relationship with the sponsor was positive and had constant feedback. The Integra IT participation in a project of this magnitude stood out for its compliance with deadlines and the delivery of information: immediate notification of grade 3 symptoms, as well as the issuance of weekly adherence reports to e-Diary and Vigilant-e so that the site could verify and act in a timely manner.
Lessons
learned
Thanks to the experience with our application in the COV002 study, the site was better prepared for the Phase III study which included thousands of subjects, as well as the management of applications.
As reported by the site, the Integra applications used for the COV002 study were very easy to use and had much more information accessible to investigators and participants, allowing timely action to be taken.
Integra IT has always provided physical support directly on site, even at the beginning of the study when the health context of the pandemic made it complicated.