Table of Contents
What is eConsent and How Does it Improve Clinical Trials?
We all recognize the vital role informed consent plays in clinical trials. These trials operate under a complex set of regulations governing how consent is obtained.
It is estimated that eConsent implementation in clinical trials reduce the time spent on consent-related tasks in trials by up to 50%, according to the article Comparative Effectiveness of eConsent: Systematic Review in PubliMed.
While paper forms remain an option, electronic informed consent (eConsent) is rapidly becoming the preferred method due to its efficiency and auditability. However, ensuring your eConsent process adheres to these regulations is essential.
In short, eConsent enhances the participant experience by:
Simplifying the consent process: Participants can access the consent document on a secure digital platform (computer, tablet, or mobile device) at their convenience.
Encouraging informed decision-making: Participants can review the information at their own pace, ask questions, and gain a clear understanding of the study before electronically signing the document.
Recommended lecture: How To Achieve A 98% EDiary Reporting Compliance Rate In Older Adults With TrialPal
Boosting enrollment rates: eConsent can streamline the enrollment process, making participation more accessible. Quick fact: A survey revealed that over 70% of patients found electronic consent more convenient than traditional paper-based methods.
Reducing paperwork: eConsent eliminates the need for physical forms, saving time and resources.
Ensuring compliance: A robust eConsent platform helps trials meet all compliance requirements.
EConsent in Clinical Trials: Features
eConsent refers to the electronic process of obtaining informed consent. Participants review the consent document on a secure platform and electronically sign it once they understand and agree to participate.
AI accelerator for multimedia content: Vieo generation, images and audios through AI. Test question generation to validate comprehension.
Digital Signature Capture: Secure electronic signatures provide a clear audit trail and demonstrate participant consent.
Bilingual Support: Catering to diverse participants with multiple language options promotes understanding and informed decision-making. (Spanish/English)
Version Control and Audit Trails: Meticulously track all eConsent forms and revisions, ensuring a clear record for audits.
User-Friendly Design: Our eConsent forms are clear, concise, and easy to navigate, regardless of a participant’s technical expertise.
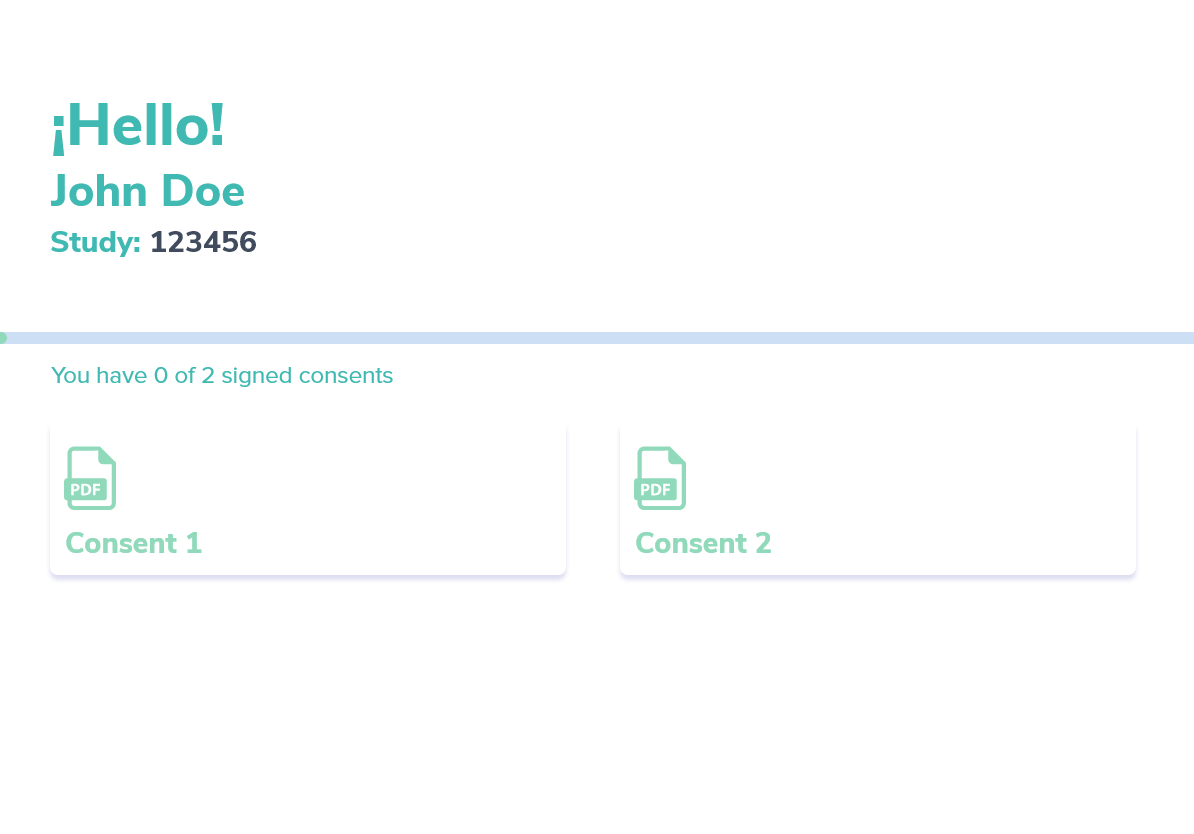

Beyond the Platform: Advantages of Integra IT's eConsent Solution
At Integra IT, we’ve taken eConsent a step further by developing a system with dynamic, multimedia-assisted informed consent information. Here are some key benefits:
Centralized Consent Management: All pending participant consents are stored and accessible within the platform, allowing participants to complete them one at a time.
Knowledge Checks: Consent statements are divided into sections with knowledge checks to ensure participants understand the information before proceeding.
Interactive Multimedia: We present all informed consent information dynamically with multimedia elements to enhance comprehension.
Streamlined Workflow: Once all sections are completed correctly, participants can electronically sign the informed consent form for final approval.
No internet-No problem: The app saves the report on the device and once it is back online all the pending data will be sent automatically.
Ready to Learn More?
Discover how our platform can simplify eConsent management and streamline your clinical trials. Fill out the form and download full brochure


