Delegation Log in Clinical Trials
Trial360 delegation log is a vital tool for research sites. It serves several crucial purposes, ensuring compliance, accountability, and efficient study conduct.
Get enhanced accountability by clearly assigning tasks and responsibilities, ensuring everyone is aware of their specific roles. This helps identify any potential issues with task execution.
Additionally, with a robust Delegation Log, research teams can effectively manage workflows as well as:
Delegation Log in Trial360 Benefits
- Facilitates smooth audits and investigations, ensuring data integrity and study quality.
- The delegation log can be used to identify training needs based on assigned responsibilities.
- Helps identify and mitigate potential risks.
- This leads to better efficiency, improved quality, and ultimately, successful clinical trials.
- Facilitates recordkeeping and updates, saving time and effort.
- Electronic records are readily accessible to authorized personnel and securely stored.
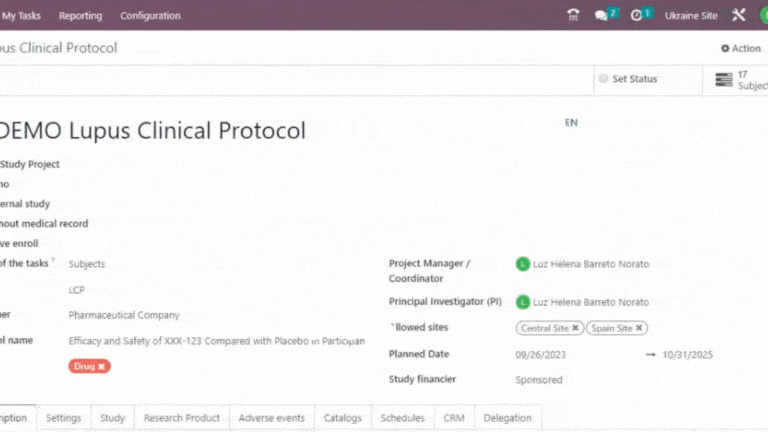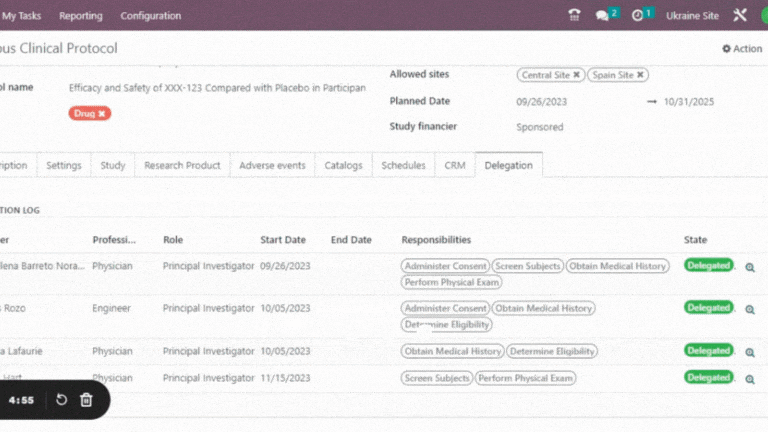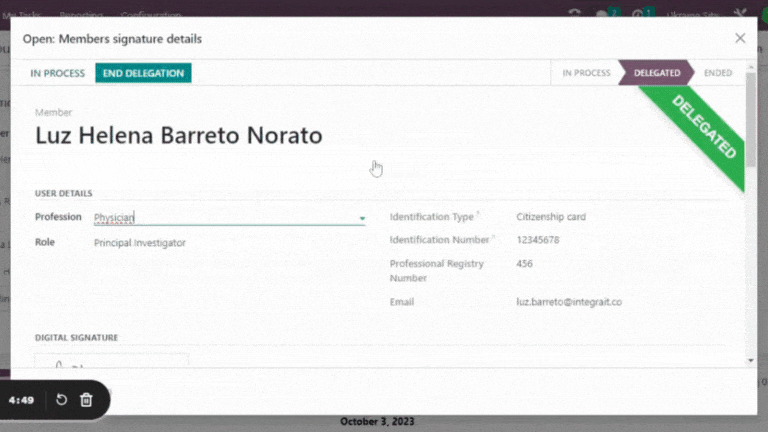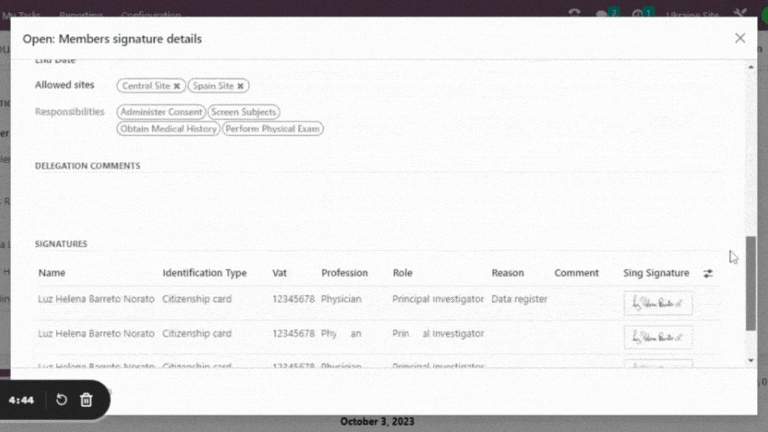
Leverage the cross-functional Trial 360 features within Quality – PI Oversight to maximize its effectiveness. Filter visits by review status or user, perform effortless searches and organize visits into groups. For added convenience, view quality review statuses directly within the visits section of the eSource/EHR.
Contact us to learn more about the Trial360 Delegation log feature.




